BrioHealth Solutions Receives FDA Approval to Begin Trial for Advanced Heart Failure Solution
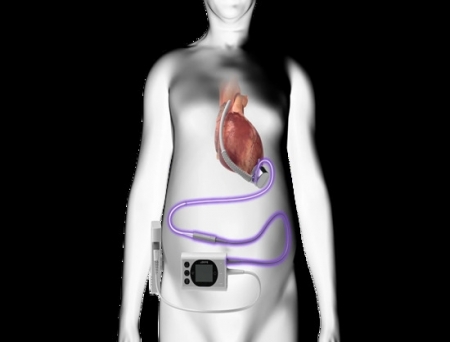
BrioHealth Solutions, Inc., a medical technology company specializing in advanced heart failure solutions, has received FDA Investigational Device Exemption (IDE) approval with conditions to initiate enrollment in its INNOVATE trial. This trial will evaluate the effectiveness and safety of the innovative BrioVAD™ System in treating refractory left ventricular heart failure.
The BrioVAD Left Ventricular Assist System offers both temporary and long-term support solutions for patients suffering from advanced heart failure. It features a novel hemocompatible blood pump with a fully magnetically levitated rotor and two small, lightweight, patient-worn components. Designed to overcome the limitations of heart failure and enhance quality of life, the BrioVAD system will be assessed in the INNOVATE trial, which is randomized relative to previously FDA-approved technology.
Francis D. Pagani, M.D., Ph.D., the Otto Gago MD Endowed Professor in Cardiac Surgery at the University of Michigan and the study's National Principal Investigator, expressed the significance of achieving FDA approval for the trial. "Achieving this stage of FDA approval for the INNOVATE trial marks an important step in providing more options to clinicians and patients seeking advanced heart failure therapies," said Dr. Pagani. "The clinical evaluation of the BrioVAD System is a significant leap forward for the entire field of heart failure."
Chen Chen, Ph.D., CEO of BrioHealth Solutions, emphasized the company's commitment to patient-centered solutions. "We are excited to have achieved this crucial milestone," remarked Dr. Chen. "Our goal is to deliver patient-centered solutions for advanced heart failure patients and their families. The innovative BrioVAD System, with its compact pump designed for hemocompatibility along with small, lightweight peripherals, has been engineered to restore patients back to active lifestyles."
Last news about this category
We use our own and third party cookies to produce statistical information and show you personalized advertising by analyzing your browsing, according to our COOKIES POLICY. If you continue visiting our Site, you accept its use.
More information: Privacy Policy

















