Clear Guide Medical Receives FDA Clearance for SCENERGY System
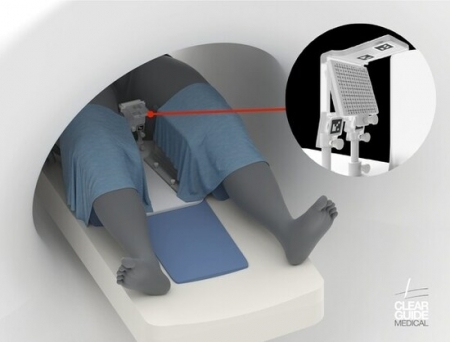
Clear Guide Medical, a medical technology innovator has announced the FDA clearance for its latest advancement, the CLEAR GUIDE SCENERGY computer-aided instrument guidance system. Alongside this milestone, the company introduces the TP Access Tool with SteriGRID™, further enhancing its suite of medical solutions.
The CLEAR GUIDE SCENERGY system represents a leap forward in image-guided diagnostic and interventional procedures, offering unparalleled accuracy and precision. By leveraging advanced features such as image fusion, instrument recognition and tracking, multi-modal markers, and target planning functionalities, the SCENERGY system empowers clinicians to perform in-suite MR guided transperineal procedures with exceptional confidence and efficacy.
"We are thrilled about the evolution of the SCENERGY product. While we always believed it would have a profound impact on cancer screenings, the clinical results for transperineal interventions have been truly remarkable," remarked Clear Guide Medical's Chief Medical Officer, Dr. Nick Karahalios.
Highlighting the company's commitment to advancing medical technologies, Clear Guide Medical's Chief Operating Officer, Patrick Duke, expresses pride in achieving FDA clearance for the SCENERGY system. "This latest FDA clearance exemplifies our commitment to advancing medical technologies for the benefit of patients and healthcare professionals," said Duke.
The FDA clearance signifies the SCENERGY system's compliance with regulatory standards, affirming its readiness for use in transperineal interventions as a new standard of care. This achievement underscores Clear Guide Medical's dedication to tailoring patented technologies to address emerging clinical needs effectively.
Specifically designed for transperineal biopsy procedures, the CLEAR GUIDE SCENERGY-TP empowers clinicians to guide biopsy needles into the prostate through the perineal skin, significantly reducing the risk of contamination associated with transrectal biopsies. According to the Mayo Clinic, this approach substantially decreases the risk of sepsis compared to transrectal biopsy methods.
Furthermore, the SCENERGY system enables the diagnosis and treatment of metastatic lesions previously inaccessible with transrectal procedures, as demonstrated in a study featured on ClinicalTrials.gov.
With the introduction of the TP Access Tool with SteriGRID™, Clear Guide Medical further enhances its offerings, providing clinicians with comprehensive solutions for improved patient care and outcomes.
Last news about this category
We use our own and third party cookies to produce statistical information and show you personalized advertising by analyzing your browsing, according to our COOKIES POLICY. If you continue visiting our Site, you accept its use.
More information: Privacy Policy

















