Galapagos, BCA to Boost Decentralized CAR-T Manufacturing Network in US
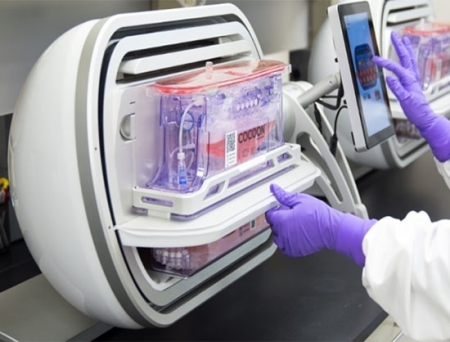
Galapagos has inked a strategic partnership with Blood Centers of America (BCA) for the decentralized manufacturing of Galapagos’ CAR-T cell therapies through BCA’s network in the US.
Under the terms of the collaboration agreement, BCA will provide access to its extensive network of over 50 community blood centers in 43 states for Good Manufacturing Practice compliant manufacturing for Galapagos’ CAR-T hemato-oncology clinical program. This agreement complements existing collaborations between Galapagos and Landmark Bio and Thermo Fisher Scientific.
Galapagos will leverage BCA’s network to initiate technology transfer to multiple sites in parallel for the decentralized manufacturing of its CAR-T product candidates, close to cancer treatment centers, while also accessing apheresis capacity at BCA sites when required. In addition, BCA will play a crucial coordinating role by supporting site initiation and onboarding to accelerate Galapagos’ efforts and ensure consistent quality.
Galapagos’ innovative, decentralized manufacturing platform could address many of the limitations that currently available CAR-T production is facing. It has the potential to offer greater speed and scalability, with the delivery of fresh, fit cells with a vein-to-vein time of seven days and the possibility for greater physician control and improved patient experience.
“Our collaboration with Blood Centers of America marks a major milestone for Galapagos’ U.S. expansion by establishing centers for support of our pivotal studies, with the potential to be used for commercial introduction. Working with BCA’s extensive nationwide network allows us to efficiently scale up decentralized CAR-T therapy manufacturing across the US, while their established infrastructure helps us harmonize operations and access apheresis capacity,” said Dr. Paul Stoffels, CEO and Chairman of the Board of Directors of Galapagos.
“This agreement complements the existing collaborations between Galapagos and Landmark Bio and Thermo Fisher Scientific and translates to a significant step forward in our aspiration to bring CAR-T therapies to more patients with a vein-to-vein time of just seven days,” Stoffels added.
“We are very pleased to partner with Galapagos in expanding their nationwide CAR-T manufacturing network,” said Delisa English, BCA Board Chair.
“Our decades of experience and advanced capabilities in regulatory-compliant biologic processing across the US will facilitate seamless technology transfer across multiple sites. We strongly believe that the combination of Galapagos’ decentralized manufacturing platform and our national cell therapy manufacturing footprint will ultimately benefit patients by providing convenient access to our local facilities for apheresis and local healthcare providers within their communities,” English added.
Last news about this category



We use our own and third party cookies to produce statistical information and show you personalized advertising by analyzing your browsing, according to our COOKIES POLICY. If you continue visiting our Site, you accept its use.
More information: Privacy Policy














